Calorimetry Problems, Thermochemistry Practice, Specific Heat Capacity, Enthalpy Fusion, Chemistry - YouTube

Chapter 17 Thermochemistry. Thermochemistry: Study of energy changes that occur during chemical reactions and changes in state Section 17.1: The flow. - ppt download

Chem 30 Formulas - 123 - Chemistry 30 – Formulas and Information Thermochemistry: Formula: - Studocu
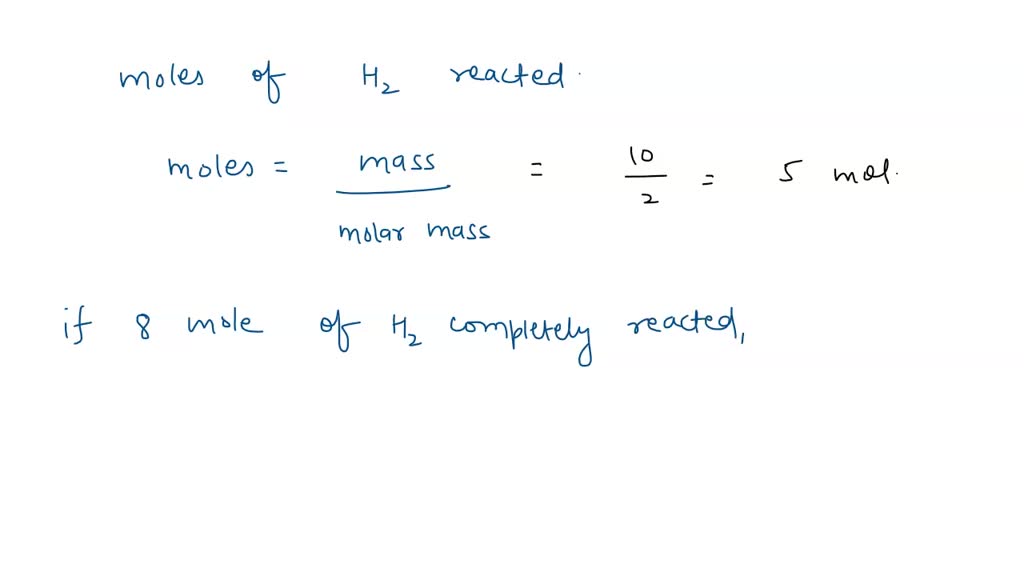
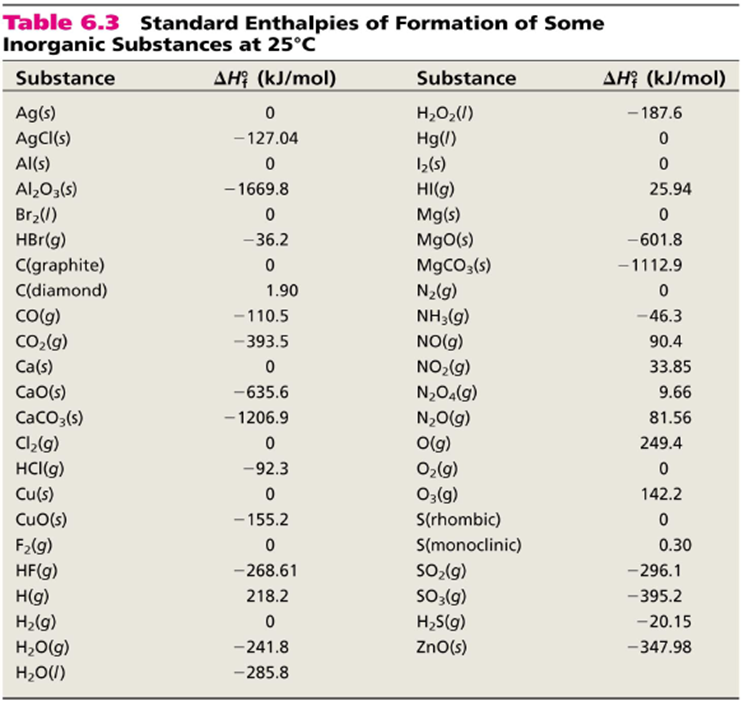
SOLVED: Consider the following thermochemical equation: S8 + 8 H2 → 8 H2S ∆rH = −161.6 kJ/mol-rxn a. Determine qrxn if 0.250 mol H2S is produced. b. Determine qrxn when 10.0 g